Altmetric Score
Dimensions
Citation
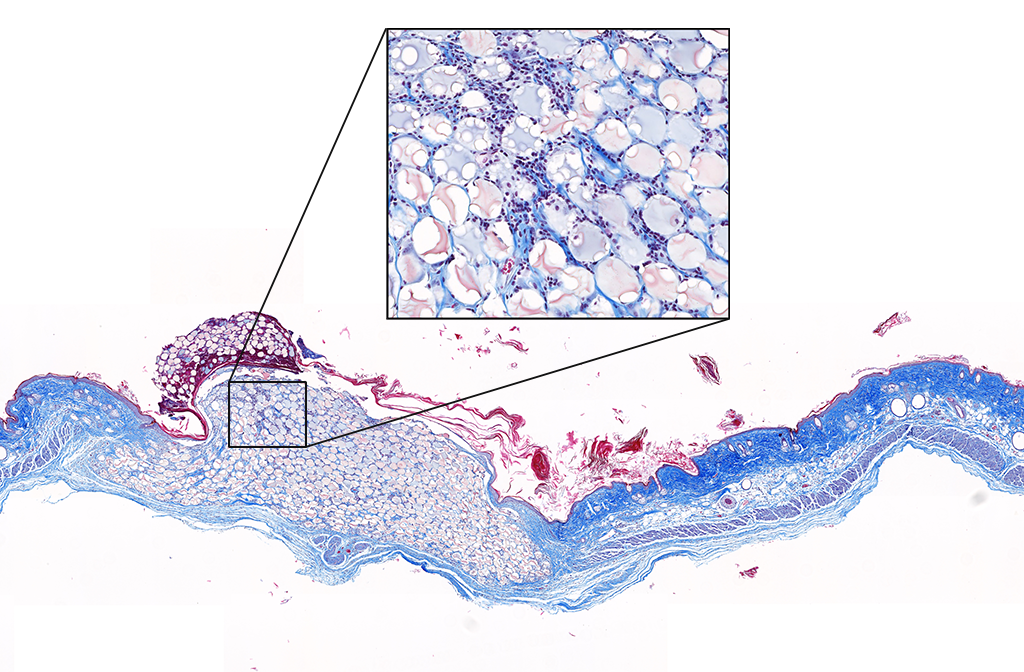
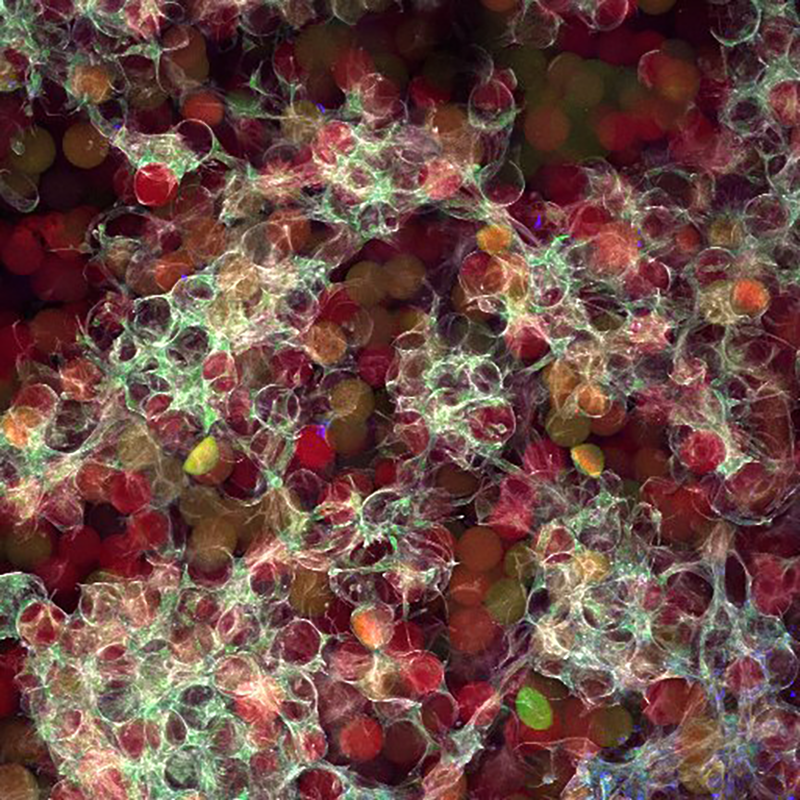
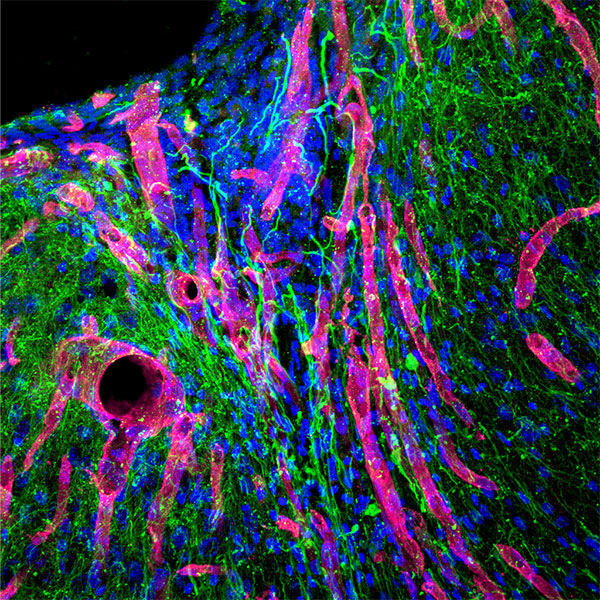
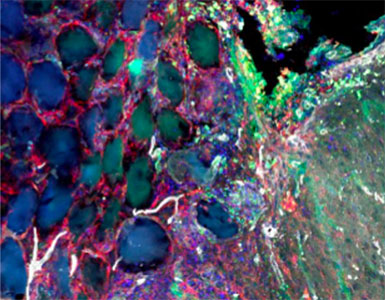
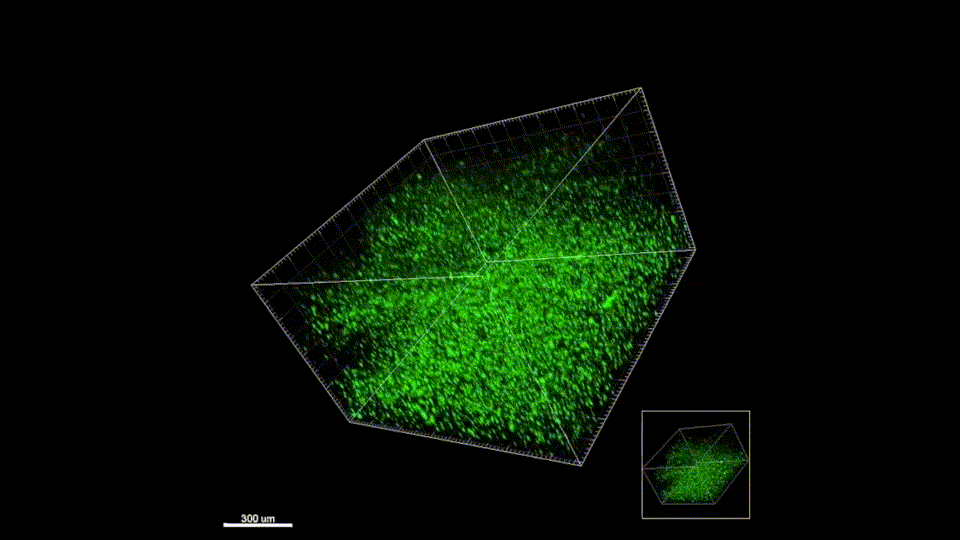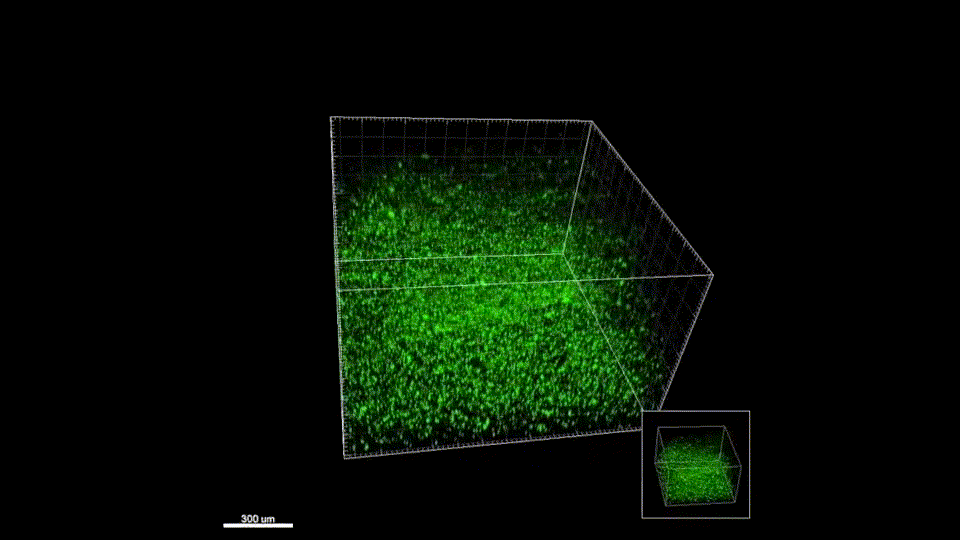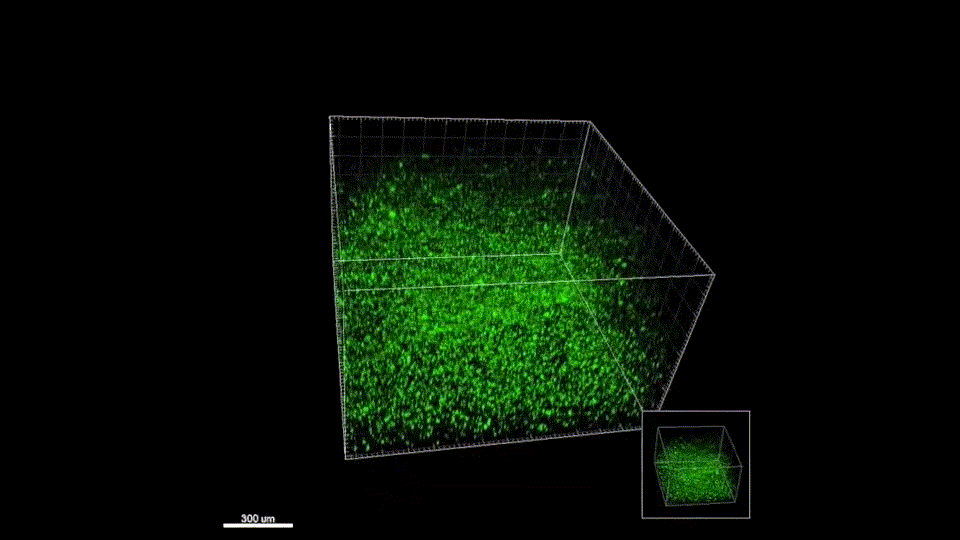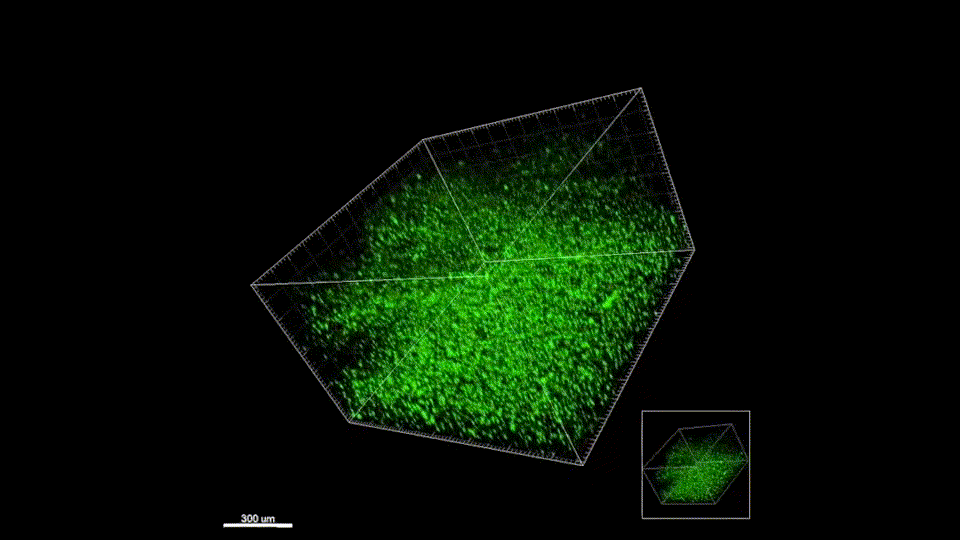
Macroporous scaffolds are being increasingly used in regenerative medicine and tissue repair. While the recently developed microporous annealed particle (MAP) scaffolds have overcome issues with injectability and in situ hydrogel formation, limitations with respect to tunability to be able to manipulate hydrogel strength and rigidity for broad applications still exist. To address these key issues, here hydrogel microparticles (HMPs) of hyaluronic acid (HA) are synthesized using the thiol-norbornene click reaction and then HMPs are subsequently annealed into a porous scaffold using the tetrazine-norbornene click reaction. This assembly method allows for straightforward tuning of bulk scaffold rigidity by varying the tetrazine to norbornene ratio, with increasing tetrazine resulting in increasing scaffold storage modulus, Young’s modulus, and maximum stress. These changes are independent of void fraction. Further incorporation of human dermal fibroblasts throughout the porous scaffold reveals the biocompatibility of this annealing strategy as well as differences in proliferation and cell-occupied volume. Finally, injection of porous HA-Tet MAP scaffolds into an ischemic stroke model shows this chemistry is biocompatible in vivo with reduced levels of inflammation and astrogliosis as previously demonstrated for other crosslinking chemistries.